FDA panel backs liraglutide for obesity
pharmafile | September 12, 2014 | News story | Manufacturing and Production, Sales and Marketing | Arena Pharmaceuticals, Belviq, Novo Nordisk, Qsymia, Victoza, Vivus, diabetes, obesity
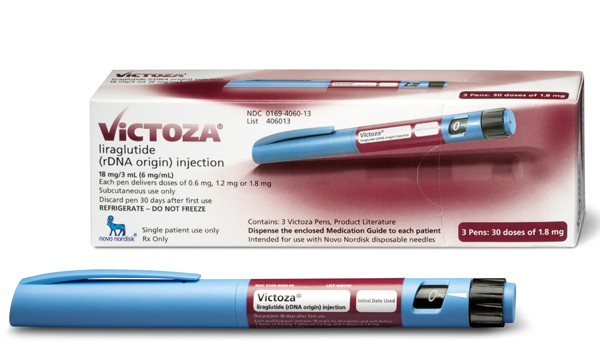
An FDA panel has voted 14-1 to allow a form of Novo Nordisk’s type 2 diabetes treatment Victoza to help obese patients lose weight.
If it gains final FDA approval (and the regulator usually follows the advice of its specialist panels) Novo says the new treatment will be called Saxenda, and is made up of a 3mg dose of liraglutide, the main ingredient in Victoza.
According to analysts it could generate an extra $1 billion in revenue for the company.
The panel’s recommendation comes from one main study where half of obese patients given a daily 3mg injection of the drug lost at least 5% of body weight, while 22% lost more than 10 per cent.
The treatment is proposed for use in patients who also have at least one other weight-related condition, such as high blood pressure.
A different dosed form of this drug is already sold under the brand name Victoza (liraglutide) for certain type 2 diabetes patients who are not able to control their blood sugar levels.
It is usually taken as an add-on to metformin or sulphonylurea when these drugs are no longer helping patients to keep their blood sugar levels down, and is the step before needing to use insulin.
It made just over $2 billion in sales last year, a growth of 27% on 2012 sales, but has faced safety concerns from regulators, predominately over its potential to increase the risk of side effects on the pancreas.
Growing market
This comes in the same week that in the US the FDA approved the fourth such obesity pill in the form of Orexigen’s Contrave, and follows the 2012 approvals of Vivus’ Qsymia (phentermine/topiramate) and Arena Pharmaceuticals’ Belviq (lorcaserin).
These were the first new obesity treatments approved in the US since Roche’s Xenical (orlistat) in 1999.
But all of these medicines have suffered a difficult path on the road to approval, with a number of FDA rejections and concerns from doctors over safety, notably the effect of these medicines on the heart.
This has led to poor sales trajectories, despite the high rate of obesity in the US. Arena reported Belviq sales of just $5.7 million in 2013, with $5.3 million of that going to its partner Eisai.
Qsymia’s sales were higher at $23.7 million in the same period, but still weak given the market potential.
This means Saxenda, should it gain approval, would be fifth such medicine to market, but analysts still expect the drug to do well given that it has been used for four years by doctors for diabetes, and has not faced the same cardiovascular safety concerns as many of its rivals.
According to the US Centers for Disease Control and Prevention, two-thirds of American adults are overweight or obese.
Furthermore, the prevalence of obesity in the US more than doubled among adults from 1980 to 2010, meaning any new – and safe – obesity drugs could be highly lucrative, although no company has yet managed to bring their drug into the blockbuster range.
Ben Adams
Related Content

FDA approves Wegovy for treatment of MASH in adults with liver fibrosis
The US Food and Drug Administration (FDA) has granted accelerated approval for Wegovy (semaglutide) as …

BioMed X and Novo Nordisk partner on oral peptide drug delivery innovation
BioMed X has announced a new collaboration with Novo Nordisk to improve oral delivery of …
Lilly reports positive results from trial of obesity candidate
Eli Lilly and Company (Lilly) has announced topline results from its phase 3 ATTAIN-1 trial, …






